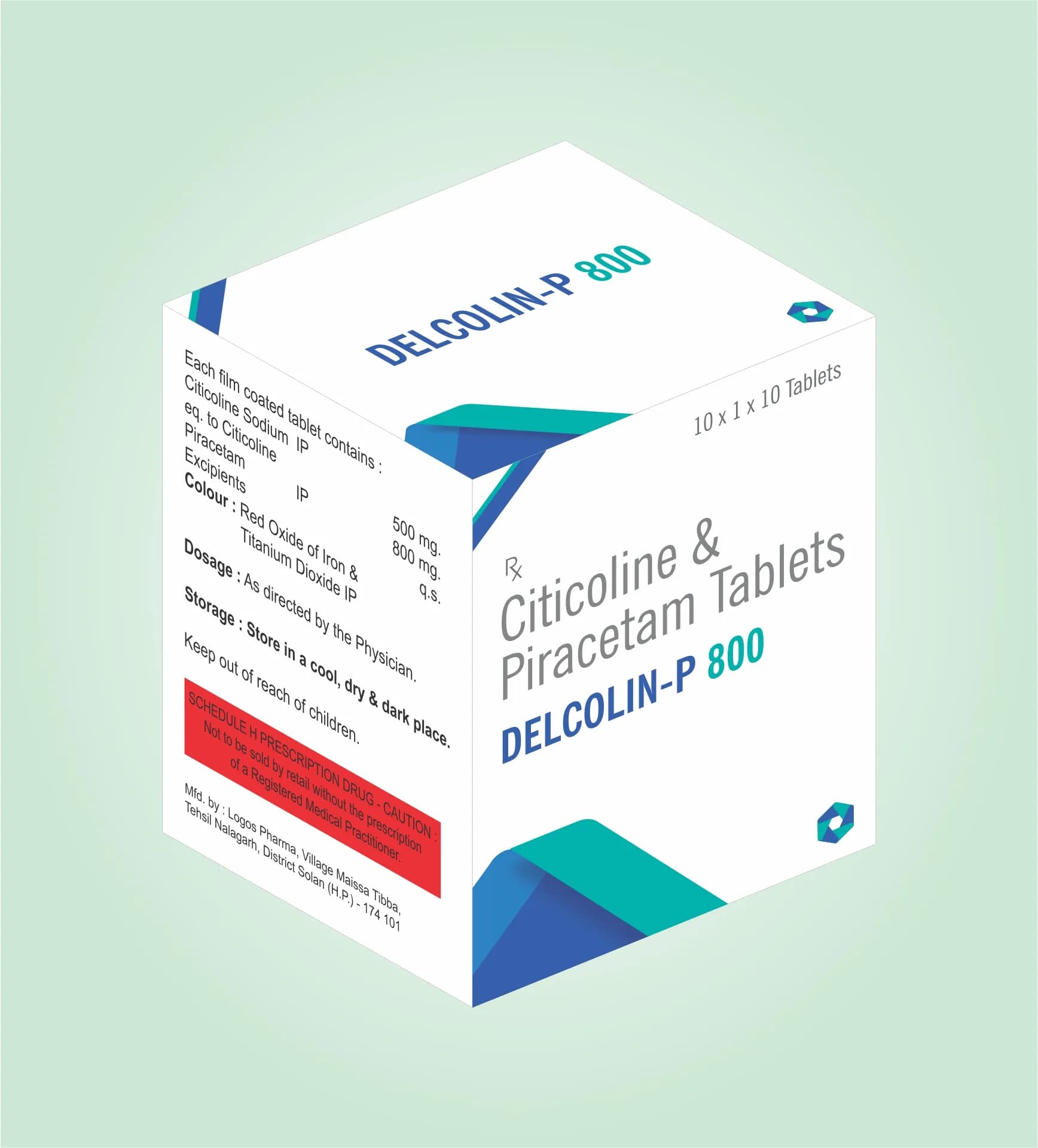
Delcolin P -800 is a dual-action formulation combining Citicoline (500mg) and Piracetam (800mg) in a single tablet, engineered for stability, bioavailability, and compatibility. The tablet formulation employs advanced excipient selection to ensure uniform drug distribution, optimal disintegration, and prolonged shelf life. Citicoline, a phospholipid derivative, is incorporated using direct compression techniques to maintain its structural integrity, while Piracetam, a nootropic compound, is blended with excipients to enhance solubility and dissolution rates. The formulation adheres to strict pharmaceutical standards, ensuring consistent dosing and minimal degradation under storage conditions. The tablet’s design balances mechanical strength with ease of swallowing, making it suitable for large-scale production and diverse therapeutic applications.
Manufacturing Capability
Delcolin P -800 is manufactured using state-of-the-art pharmaceutical infrastructure, including automated tabletting machines, high-precision blending systems, and controlled environment storage facilities. The production process complies with current Good Manufacturing Practices (cGMP), ensuring sterility, contamination control, and batch consistency. The facility is equipped with advanced quality assurance systems, including real-time monitoring of critical parameters such as temperature, humidity, and particle size distribution. Scalability is achieved through modular production lines capable of handling bulk orders while maintaining adherence to Good Manufacturing Practice (GMP) standards. Quality systems are integrated throughout the manufacturing lifecycle, from raw material sourcing to final packaging, ensuring compliance with international regulatory frameworks and pharmaceutical industry benchmarks.
Available Strengths and Packaging
Composition: Citicoline 500mg + Piracetam 800mg Tab
Packaging: 10x1x10 Alu Alu
Delcolin P -800 is available in a single strength formulation, ensuring uniform dosing for therapeutic applications. The packaging consists of 10 blister strips, each containing 1 tablet, sealed in a 10-piece aluminum-aluminum (Alu Alu) blister pack. This design provides robust protection against moisture, light, and physical damage, ensuring product stability during storage and transportation. The packaging format is optimized for efficient distribution, retail display, and compliance with global pharmaceutical packaging regulations.
Contract Manufacturing / Third Party Manufacturing
As a trusted manufacturer, Delwis Healthcare offers contract manufacturing, private label production, and third-party manufacturing services tailored to pharmaceutical buyers. Our facility supports the production of custom formulations, including combination tablets like Delcolin P -800, under brand-name or private-label agreements. We provide end-to-end solutions, from raw material sourcing to finished product packaging, ensuring compliance with regulatory standards and client specifications. Our capabilities extend to scalable production, flexible formulation adjustments, and adherence to global quality benchmarks, making us a preferred partner for manufacturers seeking to enter new markets or expand product portfolios.
Quality Assurance
Delcolin P -800 undergoes rigorous quality control systems to ensure consistency, safety, and efficacy. Each batch is subjected to comprehensive analytical validation, including high-performance liquid chromatography (HPLC), dissolution testing, and microbial limit checks. Quality assurance protocols align with International Council for Harmonisation (ICH) guidelines, United States Pharmacopeia (USP), and European Pharmacopoeia (EP) standards. Batch testing verifies active ingredient content, disintegration time, and physical properties, while stability studies confirm shelf life under accelerated and long-term conditions. Our quality systems are designed to meet the demands of global pharmaceutical markets, ensuring compliance with Good Manufacturing Practice (GMP) and regulatory requirements.
Regulatory Compliance
Delcolin P -800 is manufactured in full compliance with Good Manufacturing Practice (GMP) standards, WHO guidelines, and ISO 9001/14001 certifications. The product adheres to regulatory frameworks in the US, EU, India, and other key markets, ensuring eligibility for export and distribution. Our facility meets the requirements of the FDA, EMA, and other global regulatory bodies, with documentation and audit readiness for inspections. Compliance with ISO 14001 ensures environmental responsibility, while ISO 9001 guarantees adherence to quality management systems. These certifications position Delcolin P -800 as a globally compliant product, suitable for pharmaceutical buyers seeking to meet stringent regulatory expectations.
Global Supply Capability
Delwis Healthcare offers robust global supply capabilities, including bulk export, international distribution networks, and reliable supply chain logistics. Our facility is equipped to handle large-scale production, ensuring consistent supply for domestic and international markets. We maintain partnerships with logistics providers to facilitate seamless cross-border shipments, with compliance to customs regulations and import documentation requirements. Bulk supply capacity is supported by automated packaging lines and inventory management systems, ensuring timely delivery without compromising quality. Our supply chain is designed for resilience, with contingency planning to address disruptions and meet urgent demand from pharmaceutical distributors and exporters.
Why Choose Us
Pharmaceutical distributors, exporters, and contract manufacturing partners select Delwis Healthcare for our expertise in high-quality formulation, regulatory compliance, and scalable production. Our commitment to GMP standards, ISO certifications, and global regulatory alignment ensures that Delcolin P -800 meets the demands of diverse markets. With a focus on innovation, reliability, and customer-centric solutions, we provide tailored services to support your business objectives. Our ability to deliver bulk supply, private-label manufacturing, and export-ready products positions us as a strategic partner for companies seeking to expand their pharmaceutical portfolios.
Bulk Supply Inquiry
Delwis Healthcare offers bulk supply capabilities for Delcolin P -800. For inquiries regarding large-scale orders, please contact us at info@delwishealthcare.com.
FAQs
What manufacturing standards does Delcolin P -800 adhere to?
Delcolin P -800 is manufactured under cGMP guidelines, with compliance to WHO, FDA, and EMA regulations, ensuring global market readiness.
Can Delcolin P -800 be produced in different strengths?
While Delcolin P -800 is currently available in a single strength (Citicoline 500mg + Piracetam 800mg), custom formulations can be developed based on client specifications.
What is the shelf life of Delcolin P -800?
The product has a shelf life of 24 months when stored under recommended conditions (cool, dry, and protected from light).
Does Delwis Healthcare offer private-label manufacturing?
Yes, we provide private-label manufacturing services, enabling clients to brand and distribute products under their own labels.
What packaging options are available for Delcolin P -800?
The product is available in 10x1x10 Alu Alu blister packs, designed for stability, tamper resistance, and compliance with global packaging regulations.
How does Delwis Healthcare ensure quality control?
Each batch undergoes rigorous testing, including HPLC, dissolution, and microbial analysis, with compliance to ICH, USP, and EP standards.
Can Delcolin P -800 be exported to international markets?
Yes, our regulatory compliance and quality assurance systems ensure eligibility for export to over 150 countries.
What is the lead time for bulk orders?
Lead times vary based on order size and production capacity, but we prioritize timely delivery with a minimum turnaround of 4–6 weeks for standard orders.
Does Delwis Healthcare support contract manufacturing for other products?
Yes, we offer contract manufacturing services for a wide range of pharmaceutical products, including combination tablets and customized formulations.
How can I inquire about bulk supply?
For bulk supply inquiries, please contact us at **info@delwishealthcare.com**. Our team will provide detailed pricing, specifications, and supply timelines.